Popular

Innovation examples
HealthToxicology
Zebrafish in toxicity testing
Zebrafish are increasingly recognised as a useful model for toxicity testing of chemical substances. Testing strategies are becoming more based on mechanisms of toxicity structured in adverse outcome pathways describing the chain of events leading to toxicity or disease. Using a battery of dedicated in vitro and in silico assays, insight can be gained in how exposure leads to disease. For certain diseases it is known that toxicity relies on the interaction between different organs and cell types, which requires research on whole organisms in addition to simple in vitro models. The zebrafish is considered a valuable whole organism model in a mechanism-based testing strategy. At RIVM, the zebrafish embryo model is used for testing the effect of chemical substances on several adverse outcomes and diseases.
For more information see: https://ehp.niehs.nih.gov/doi/10.1289/EHP9888; https://doi.org/10.3390/ijerph18136717; www.linkedin.com/in/harm-heusinkveld
Expert interviews
ToxicologyIn silicoPolicy
Tox 21: A New Way to Evaluate Chemical Safety and Assess Risk
Tox21 is a US federal research collaboration focused on driving the evolution of Toxicology in the 21st Century by developing methods to rapidly and efficiently evaluate the safety of commercial chemicals, pesticides, food additives/contaminants, and medical products. The goals of Tox21 are to (1) identify mechanisms of chemically-induced biological activity; (2) prioritize chemicals for more extensive testing; and (3) develop more relevant and predictive models of in vivo toxicological responses.

Innovation examples
HealthIn vitroOrgan-on-Chip
An iPSC-derived blood-brain barrier to model neurodegeneration
The blood-brain barrier is a layer of cells that protects our brain from harmful compounds. However, due to this tight barrier, many drugs to treat neurological diseases cannot enter the brain either.
There are currently no good models to test these types of drugs. Henrique Nogueira Pinto is a PhD candidate at the Vrije Universiteit in Amsterdam. He is developing a blood-brain barrier model coupled to mini-brains. With this model, he aims to more reliably test how drugs can be transported over the blood-brain barrier and what their effect on the brain is.
Click on the info button for the full version of the video. Click here (https://fluidsbarrierscns.biomedcentral.com/articles/10.1186/s12987-022-00316-0#Sec3) for a review of the current status of in vitro models for the blood-brain barrier.

Innovation examples
ToxicologyIn vitroOrgan-on-Chip
Cartilage-on-a-chip for studying joint degenerative diseases
Carlo Alberto Paggi is currently a PhD candidate at the University of Twente in the research group of Prof. Marcel Karperien and Prof. Séverine Le Gac. Karperien’s lab focus on the biological aspects of osteoarthritic research while Le Gac’s specialize in organ-on-chip development. The project of Carlo Alberto is developing a joint-on-chip platform to create a reliable in vitro model to study disease progression in osteo- or rheumatoid arthritis. The model combines different organ-on-chips aimed at replicating each a tissue around the joint such as cartilage, bone and ligaments. This new technology focuses on better reproducing human models and at substituting the use of animal models for drug research. If you want to know something more about the project and the groups, you can follow the link in the video.
Carlo Paggi was nominated for the Hugo van Poelgeest prize for his research on a cartilage-on-a-chip model to study joint degenerative diseases
Karperien’s lab of Developmental Bioengineering: https://www.utwente.nl/en/tnw/dbe/
Le Gac’s lab of Applied Microfluidics for BioEngineering Research: http://www.severinelegac.com/
Linkedin: https://www.linkedin.com/in/carlo-alberto-paggi-76500b135/
Meetings & conferences
HealthIn vitroAdvanced
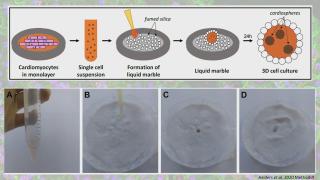
Liquid marbles for cardiac organoids development
Advances in three-dimensional (3D) culture techniques have shown several advantages over 2D cultures, especially by more accurately mimicking the in vivo environment. This has led to improved reproducibility and reliability of experimental results, which are important criteria in disease modelling and toxicity testing. Induced pluripotent stem cells (iPSC) provide an unlimited source for the derivation of all cell types of the adult body, including cardiomyocytes. To improve the current culture methods for multicellular cardiac spheroids, such as the hanging drop method, we explored the use of hydrophobic powders. Fumed silica nanoparticles can be used to encapsulate liquid drops, which could serve as a microenvironment for cell cultures. This microbioreactor stimulates cell coalescence and 3D aggregation while providing optimal gas exchange between the interior and the surrounding environment. Moreover, the properties of liquid marble microbioreactors render them ideal for co-culture experiments. This liquid marble technique has been previously explored and optimized for other cell types. Here we describe a protocol that allows for the derivation of functional cardiac mini organoids, consisting of co-cultured cardiomyocytes and cardiac fibroblasts. These cardiospheres can be valuable for modelling cardiac diseases in vitro and assessing cell interactions to decipher disease mechanisms.
Lab website: https://www.medicalcellbiologylab.com/
Contact: https://www.researchgate.net/profile/Jeffrey-Aalders
RE-place database: https://www.re-place.be/method/liquid-marbles-cost-effective-platform-generate-cardiospheres-co-cultured-cardiomyocytes-and

Innovation examples
HealthToxicologyIn silico
AI agents for safer science: How AI is Changing Chemical Risk Assessment
This video introduces a novel approach to chemical safety, where intelligent digital agents guided by large language models support scientists in making faster, more transparent decisions. By automating complex workflows and integrating tools like the OECD QSAR Toolbox, these agentic systems help prioritise research, reduce reliance on animal testing, and pave the way for safer, more sustainable innovation.
Innovation examples
HealthToxicologyIn vitro
Developmental neurotoxicity testing using stem cells
Children should grow up in a safe and healthy environment. Disruption of brain development may have enormous impact on future life and might result in disorders such as ADHD or cognitive decline. The effect of compound exposure on the developing brain is largely unknown, since in the current regulatory test procedures in experimental animals effects on the brain are rarely investigated and human relevance of these animal models is under debate.
Researchers at RIVM are developing a cell model based on human stem cells that mimics a small part of the developing brain. This method is human-relevant, animal-free, and based on mechanistic knowledge of human biology and physiology of brain development. The model can be an important component in a testing strategy to test the safety of chemicals and pharmaceuticals on the developing brain.

Innovation examples
HealthIn vitro
Immortalized human cells to model atrial fibrillation in vitro
Niels Harlaar is a PhD Candidate at the Laboratory of Experimental Cardiology at the Leiden University Medical Center. Here, under the supervison of prof. dr. D.A. Pijnappels and dr. A.A.F. de Vries, he focusses on the conditional immortalization of human atrial cardiomyocytes for (among many other applications) in vitro modelling of atrial fibrillation. He has successfully generated, characterized and applied this technique of these conditionally immortalized human atrial myocyte lines to model atrial fibrillation in vitro.
Niels won the Hugo van Poelgeest prize 2022 for excellent research to replace animal testing.
Click here (https://hartlongcentrum.nl/research/laboratory-of-experimental-cardiology/) for more information on the Laboratory of Experimental Cardiology.

Questions
HelpathonsEducation
Helpathon #3 - Can you help Daniela?
Daniela Salvatori calls for a Helpathon! She invites you to help her and TPI Utrecht to create a unique master course for animal free innovation. You can sign in for this Helpathon here: tpihelpathon.nl. Online, 18th - 19th of June 2020.

Innovation examples
ToxicologyIn vitroOrgan-on-Chip
Human pluripotent stem cell derived cardiomyocytes for disease modelling and drug discovery
Berend van Meer did his PhD research in the research group of prof. Christine Mummery at the department of Anatomy and Embryology of the Leiden University Medical Center. In this group, human pluripotent stem cell derived (Organ-on-Chip) models are being developed, mostly cardiovascular models. The work of Berend aimed to understand how well these stem cell based cardiac models can predict the effect of (well-known) drug therapies in patients. Importantly, the outcomes of the experiments were compared to very similar measurements in rabbit heart muscle cells. And while animal models predicted less than 70% correctly, the human stem cell based models predicted almost 80% of the expected effects correctly. The research contributes to understanding the relevance of stem cell based models and strengthens the confidence regulators and pharmaceutical companies have in such models as animal alternatives in the drug development pipeline.
Berend van Meer has won the Hugo van Poelgeest prize 2020 for his research on human pluripotent stem cell derived cardiomyocytes for disease modelling and drug discovery.
Christine Mummery's lab on Heart on Chip, Disease modeling and toxicity: https://www.lumc.nl/org/anatomie-embryologie/research/902040935402533/
Meetings & conferences
HealthToxicologyIn vitroAdvanced
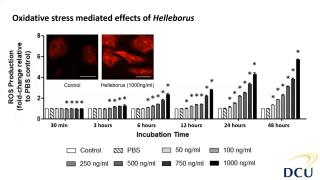
Characterisation and cytotoxicity assessment of Helleborus with NAMs
Helleborus sp. is a member of the Ranunculaceae family, and are small, perennial herbs common in Central and Southern Europe and Asia. Their distribution in Europe elevated their position in therapeutic remedies since the ancient time and mythology. Due to their potent and rich extracts from their roots, hellebores have been used in traditional and folklore remedies as they present rich sources in glycosides. Mainly, these plants have exhibited cathartic, anthelmintic and other beneficial aspects to treat diseases, however, hellebores have also been known for their adverse and poisonous aspects. It is also because of their cytotoxic aspect that these species have also been explored as alternative approaches to cancer treatment and are mainly reported as sporadic patient cases in literature. In this study, we first focused on the phytochemical characterisation of Helleborus odorus subsp. cyclophyllus combining biochemical assays and a detailed characterisation of its antioxidant and antibacterial properties. Furthermore, regarding its toxic potential, we explored the cytotoxic toxic properties and the mechanisms of toxicity mediated effects using in vitro cell systems primary human aortic endothelial cells (HAECs). HAECs are useful for studying vascular diseases such as thrombosis, atherosclerosis, and hypertension as well as for stent-graft compatibility testing and within the 3Rs principles, avoiding animals in these studies. Results showed the cytotoxic and reactive oxygen species potential of Helleborus extract in dose and time dependent manner. Further investigation (not shown here) revealed more mechanistic effects relevant to inhibition of proliferation.
Contact: https://www.researchgate.net/profile/Anna-Michalaki

Projects and initiatives
HealthIn vitroOrgan-on-Chip
SCREENED: developing 3D thyroid models
The European collaborative project SCREENED aims to develop three-dimensional (3D) cell-based in vitro tests to better characterize the effects of endocrine disruptors (EDs) on thyroid gland function. This method will overcome the limitations of existing tests, being more sensitive at low doses of exposure to chemicals, and enabling the prediction of their toxicity on human health in a sex-specific manner. The ambition of the SCREENED project is that these new 3D in vitro tests, as well as the increased knowledge about adverse reactions after exposure to EDs, will be used for regulatory purposes, ultimately to improve human health.